India’s vaccine drive will need 100,000 fridges, 11,500 refrigerated trucks, a cold-chain evolution

Gubba cold storage has been preserving perishables since 33 years through innovation, science and technology. Today, we are absolutely geared up to fulfil the crucial requirement of the nation- Preservation of Covid-19 vaccine. In that context, ET Prime has approached Mr. Gubba Kiran, CEO, Gubba Cold Storage for his views on supply chain scenario in India to handle the distribution of the covid-19 vaccine. Below is the article with his opinion on the subject.
Adar Poonawala has a pertinent question for the health ministry.
The CEO of Pune-based Serum Institute of India, which looks to manufacture about 1 billion doses of the Covid-19 vaccine being jointly developed by Oxford University and AstraZeneca, tweeted on September 26: “Quick question; will the government of India have [INR] 80,000 crore available, over the next one year? Because that’s what @MoHFW_IN needs, to buy and distribute the vaccine to everyone in India. This is the next concerning challenge we need to tackle.”
Poonawala just hit the nail on the head.
India’s fight against the wily virus won’t just end with getting a vaccine. It will rather open another front. Putting in place an apt cold-chain infrastructure — warehousing and transportation — that can handle the varying temperature requirements of different Covid-19 vaccines will be a litmus test.
To be sure, not all corona vaccines will need sub-zero temperatures. The in-the-works Oxford-AstraZenca vaccine is protein-based and may not need to be frozen but refrigerated between 2 and 8 degree centigrade. But two of the world’s leading vaccine contenders, Moderna and Pfizer-BioNTech, need more than refrigeration. Moderna’s vaccine needs to be shipped and stored at -20 degree centigrade. The Pfizer-BioNTech one needs to be kept at -70 degree centigrade.
The Hyderabad-based Dr. Reddy’s Laboratories has been in talks to distribute 100 million doses of Russia’s Sputnik V vaccine once final stages of human trials and regulatory approvals are in place. Sputnik V may require storage conditions of up to -18 degree centigrade. Some of these vaccines may need two shots after an interval of 21 days.
This makes transportation of vaccines a logistics exercise that will require a robust cold-chain infrastructure. with the health ministry’s plans to procure about 400 million- 500 million vaccine doses and immunise about 200 million-250 million people by July 2021, India’s cold-chain infrastructure will have to be ready within a tight deadline.
So how does India’s cold-chain infrastructure look as of now?
According to data from the National Centre for Cold Chain Development, as of 2018, India had a total cold-storage capacity of 30 million-35 million metric tonne– one of the world’s largest. Looks quite neat, right? The real puzzle, however, lies somewhere else.
Numbers alone don’t paint the right picture
Industry insiders say there may be about 10,000-odd cold-chain providers in the country but only about 5% of them are significant players with a capacity of up to 5,000 tonne. However, very few are pharma-compliant as per WHO guidelines. “If you ask them if they are WHO- GDP (good distribution practices) certified, 9,700 of them will say they don’t know about it,” says an industry insider.
A handful of big names, including Snowman Logistics, Gati Kausar, Kool-ex, DHL, ColdStar Logistics, MJ Logistics, Gubba Cold Storage, Kuehne + Nagel among others operate in this segment.
A bulk of the cold storage available in the country is used for agricultural produce, including potatoes and chillies. Most large cold-storage companies, including market leaders like Snowman Logistics, have about 10% of their business volume coming from pharmaceutical storage.
On the transportation side, India reportedly had more than 127,00 reefer or refrigerated vehicles as of 2018.
One of the biggest challenges in cold-chain management is lack of visibility or traceability of shipments. Vaccine logistics will require real-time and end-to-end monitoring of consignments during transit as well at the storage facilities. Lack of real-time temperature tracking of cold-chain vehicles and delays in pickups, drops, loading, and unloading of consignments can spoil the vaccines.
The Union health ministry has announced the use of Electronic Vaccine Intelligence Network (eVIN), a technological solution for immunisation supply-chain systems across the country. The eVIN system will monitor vaccine stocks and storage temperature in real-time at multiple locations across the country. It has reached 32 states and union territories and will soon be rolled out in Andaman and Nicobar Islands, Chandigarh, Ladakh, and Sikkim.
But experts say India doesn’t have large -80 degree centigrade cold-storage or transportation facilities and vaccines that need these temperature specifications are unthinkable as of now. Even the -20 degree centigrade temperature requirement will be a challenge because India does not have sizeable -20 degree cold-storage facilities. Products like icecream and meat are stored at -20 degree deep freezers. But if this existing infrastructure is to be utilised for vaccines, it needs to be turned into pharma-compliant WHO’s GDP-certified storage facility.
Vaccine preservation cannot be done in a conventional facility. It has to be done in a world-class facility, which is single door, rack supported, has an automated handling system and a data logger to measure and store a range of temperature readings.

The challenge of capacity and last-mile delivery
Most of the Covid-19 vaccine production is expected to happen across three regions – Telangana, Andhra Pradesh, Pune (Serum Institute), and Gujarat (Zydus Cadila).
Although there is no clarity yet in terms of what quantities of the vaccine will be stored and transported, two theories are floating around as per insiders. One, they may come as 2ml single-shot vials. Two, they may be 50ml vials carrying 25 calibrated doses each.
“A filled-up 2ml vial may weigh 200 gms. Each reefer truck, which has close to 7 tonne capacity, can carry close to 40,000 to 50,000 such vials. For 400 million vials with each weighing 200 gms, we need 30,000 tonne of transportation capacity, translating into 11,500 vehicles,” says JPN Singh, managing director, Scorpion Group.
These reefer vehicles will be needed for primary distribution alone — from pharma companies to the government’s primary health centres.
The distribution model for the Covid-19 vaccine will be very much different from the conventional pharma ecosystem because vaccine distribution is expected to be a government-controlled exercise.
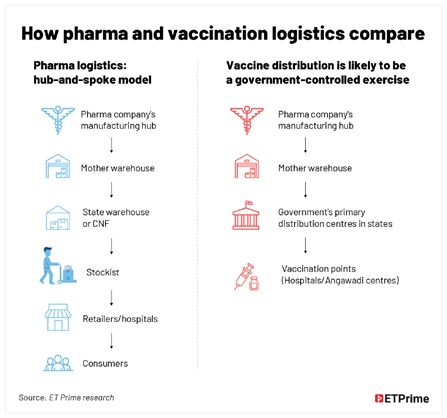
Availability of cold-chain vehicles is not a big challenge during the primary distribution from pharma companies’ manufacturing facilities to the mother warehouse. But large storage points may be a constraint at the secondary distribution points. Going further down to thousands of vaccination points across the states may be challenging in terms of storage as well as transportation. “It is easy to arrange a cold-chain vehicle from Hyderabad to Patna. But getting a cold-chain vehicle from Patna to Purnia, Muzaarpur, or Siwan would be a challenge,” says Singh. His logistics company has been delivering medicines and vaccines to the government’s Anganwadi centres, largely for pregnant women and children.
“It is more of a last-mile challenge and that needs to be worked backwards from the point of administering the vaccines,” says Pawanexh Kohli, founding CEO at India’s National Centre for Cold Chain Development. “As far as last-mile logistics is concerned, a city like Delhi-NCR which has a population of around 35 million, may need 4,000 vaccine-administering points. And each of these points will need a vaccine refrigerator to keep the vaccine at 2 to 8 degrees. So, we will have to leverage other assets or maybe create new assets, and these assets may have to be rotated across different regions, if vaccination across the country has to happen in phases. We may need more than 100,000 fridges to do the same,” he elaborates.
This is far bigger than polio vaccination
For Covid-19 vaccine distribution, the government plans to leverage the Universal Immunisation Programme (UIP). The UIP plan for 2018-22 has a vast network of 27,000 cold chain points, about 750 of these are located at district levels and further down. However, UIP, even though it is the world’s largest immunisation programme, may have limitations for Covid-19 vaccination. First, it is designed to administer vaccines largely to about 60 million people annually, largely newborns and pregnant women. Covid-19 vaccination is a 25 times bigger exercise to be executed in less than a fraction of the time — say three to four years. The polio-eradication drive has taken decades. Two, polio vaccines can be carried in a 200ml bottle and administered orally via droppers to each child. Covid-19 vaccines will have to be administered in injection form and carried in vials of 2ml each, supposedly. So, their handling will be very different.
Three, each of these 27,000 cold rooms provide normal refrigeration at 2 degree centigrade to -8 degree centigrade and have a capacity of 10 cubic metres to 15 cubic metres. For a much bigger immunisation drive, more such refrigeration points will be needed at the last mile. Amidst the shortage of refrigerating units, companies may employ the good old dry-ice (solid carbon dioxide), which sublimates at -78.5 degree Celsius, as a refrigerating medium. However, experts say dry-ice won’t be a feasible option for maintaining temperatures in the minus range, as may be the requirement of some vaccines. Also, ample availability and production of dry-ice across the country will have to be looked into. The last leg of delivery of vaccines such as the polio vaccine is managed by thermocol-packed dry-ice containers, which can maintain temperature for 24 to 48 hours. Thereafter it is stored in refrigerators at the primary health centres. There are no clear answers yet whether similar things will work for Covid-19 vaccines.
Private players need clarity
The government needs to rope in private players before it is too late. The Centre has directed states to make a robust plan for cold storage and transportation of vaccines by October 15. “The government of India must talk to private players who have the ability to scale and do a joint programme with them,” says Rahul Agarwal, managing director at cold-chain logistics company Kool-ex, which recently tied up with leading warehousing player Indospace to build pharma-specific warehouses. Private players ET Prime spoke to are willing to allocate resources and add capacity, but as of now many are waiting for clear directives from the government and pharma companies. “As of now, logistics companies are trying to assess capacities available in the market. Large logistics companies are trying to map the cold-chain companies in the country. But no one has started investing in capacity because no one knows what capacity they will have to build and at what location. That clarity is yet to come,” says Sunil Nair, CEO at Snowman Logistics, India’s largest cold-chain player with a total capacity of 120,000 tonne of storage across 15 cities and 300 reefer trucks. The company has been handling the primary storage of vaccines for influenza and typhoid, active pharmaceutical ingredients (APIs), and other specialty chemicals. The company’s facilities are equipped to handle temperature requirements of -25 to +2 to 5 degree centigrade and at present 85% of its total capacity is utilised. If the need arises, Snowman says it is ready to create more capacity or collaborate with other partners or create more space within existing infrastructure by optimising its inventory.
The company says its facilities are meant for storage of food as well as pharma products and these can be interchanged by handling temperature requirement, cleaning, and fumigation, which takes 24 hours. But interchangeability between food and pharma is not going to be easy since pharma is not more than 10%- 20% of most cold-chain companies’ business. The large cold-chain companies in India are keeping their fingers crossed, as they are yet to get any indication on the requirements and by when they will need to get enough cold-chain infrastructure ready. While some have begun talks with pharma companies, they are yet to sign on the dotted line.
“We can make way for vaccines by pushing out fruits or meat products or ice-cream, but getting these set-ups ready for vaccines may take about three to four months,” says Gubba Kiran, CEO at Telangana-based Gubba Cold Storage, one of India’s largest cold-chain players with a total capacity of 9,000 pallets for its pharma vertical.
It is going to be a highly capital-intensive exercise. Setting up a 6,000 pallet capacity needs 3 acre land with a total investment of about INR30 crore. Also, about a year is needed for construction.
There is one more factor playing out in the minds of the cold-chain companies. Building new infrastructure requires capital but the vaccine exercise might not last for long and the return on investment may take seven to eight years. Also, it is not an easy thing to replace the existing food clients with the ones from pharma. “It is like riding a tiger while still managing your business,” says Kiran.
Mumbai-based Kool-ex Cold Chain is designing a portable walk-in cooler, which can be transported from manufacturing plants to pharma companies’ mother warehouses and state warehouses. These walk-in coolers can maintain temperature at -20 degree centigrade. “Pharma companies may not be ready to make heavy capex in these facilities, especially since companies will see a surge in demand for six months or so and then things will settle down,” says Agarwal. His solution, if comes into practice, can be deployed on a rental basis and can store about 100,000 vaccine vials in each box.
The bottom line
India’s initial fight against the coronavirus outbreak offers some important lessons. As per a white paper released by DHL and McKinsey, inefficient warehousing has resulted in damages in supplies of PPEs and even hand sanitisers, which, because of their alcohol content, need careful handling for long-term storage. Due to the suddenness of the event, warehouses, not actually designated for life-science products, were used for medical supplies, including PPE. Many of the warehouses did not meet the necessary conditions such as temperature and humidity controls, resulting in damaged or deteriorated supplies. Waste of vaccines due to failure in adhering to handling protocols is a global problem as reported by WHO. In India too, vaccine wastage is upwards of 10%. If India doesn’t want to repeat the initial mistakes and deliver the Covid-19 vaccines to the last mile effectively, almost every mode of logistics has to be used whether the consignment arrives at the airports or the vaccine is indigenously manufactured. As per the report by DPDHL, two thirds of the global population are unlikely to have easy access to any Covid-19 vaccine preserved at freezing temperatures. The lower the temperature requirement, the more difficult and costly the entire exercise will become. Some companies like US-based courier giants UPS are working on a couple of freezer farms, each exceeding the area of a football field and can store millions of doses of Covid-19 vaccines at -70 to -80 degree centigrade. In India, primary distribution has to happen to states followed by secondary distribution to cities and talukas, and then to small hospitals before finally being distributed. There has to be adequate cold-storage facilities plus transportation via temperature-controlled vehicles for the vaccines to retain their potency. Complete end-to-end visibility along the entire supply chain will be key. Any slippages or lack of coordination in cold chain capabilities can be disastrous.