Novartis, Propeller Health co-package new asthma treatment
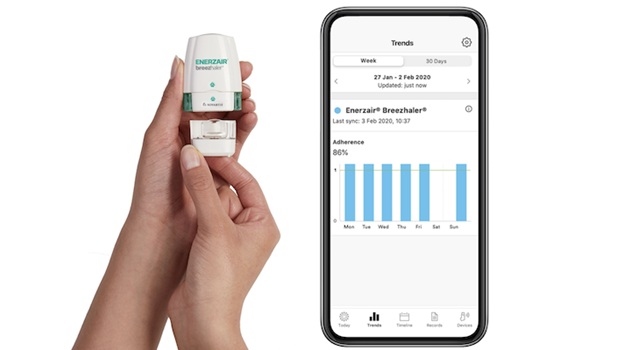
Novartis and respiratory health company Propeller Health, a ResMed subsidiary, have teamed up to co-package the latter’s digital health platform with the pharma giant’s new asthma medication Enerzair Breezhaler.
This news come shortly after the product landed European Commission clearance. The co-packaged product will be launching in the EU during 2020. However, it is not yet available in the U.S.
“I think today’s news is an important one, because I think it’s the first time we’ve ever seen a respiratory medicine be co-packaged and be available for co-prescription alongside a tool like Propeller, a digital health platform,” David Van Sickle, cofounder and CEO of Propeller Health, told MobiHealthNews. “We believe, and have for a while, that this is going to make it easier for patients and their physicians to benefit from digital health and improve their treatment plan. It’s really an important day for us, because we believe it is going accelerate the adoption of digital respiratory health in Europe and help spur other innovative approaches to combining medicines plus digital across the industry.”
Propeller’s sensor is able to attach to the Enerzair Breezhaler inhaler, where it can collect medication data and send that information to a corresponding app. The tool works both as an adherence measure that gives the patient reminders to take their medication, and as a means of collecting information about the patient’s condition.
Van Sickle noted that, historically, patients were responsible for recording their own symptoms and exacerbations – which can be difficult to remember over a long period of time. He pitches the tech as a way for patients to keep track better.
“What digital can do is provide a vital sign that is more objective and timelier. A patient and physician together can better understand whether treatments really control symptoms,” he said. “But with Propeller they are able to get real-time data about the burden of that disease, and that is essentially a marker for how well they are doing – if they are using too much of a [quick relief inhaler] and they need a control medication added, or they need a higher dose of it, or have another thing going on. It opens up a whole new prospective panel of patients and can help providers identify who needs more attention.”
WHY IT MATTERS
Asthma is a common condition and impacts 7.7% of U.S. adults and 7.5% of children in the country, according to the CDC. Doctors may prescribe a combination of quick -relief inhalers and long-term-control medication, according to the Mayo Clinic.
Novartis and Propeller are pitching this new co-packaged product as a way to help manage the symptoms of the disease.
“Historically the majority of people who have had asthma have not had their symptoms under control,” Van Sickle said, “I mean, given that we have great medicine and all that we’ve learned about the disease. It’s really been a frustrating gap, and we should have been able to help people control these diseases much more than we have as a population.”
THE LARGER TREND
Propeller and Novartis have a long history. In 2017, the two announced a collaboration to develop a custom add-on sensor for Novartis’ Breezhaler inhaler, which is used for Novartis’s portfolio of COPD treatments.
Propeller is no stranger to the pharma space. In May it announced that its sensor and app landed FDA 510(K) clearance for use with AstraZeneca’s Symbicort inhaler for asthma and COPD. It has previously made deals with Boehringer Ingelheim and GSK, and has been used in a number of clinical trials.
Novartis has a history of working with digital health companies. Perhaps most notable was its relationship with digital therapeutic Pear. Novartis’ Sandoz was working with the startup to commercialize its FDA-cleared addiction-focused products reSET and reSET-O. Last fall, the pharma giant announced that it was handing sole responsibility of the commercialization back to Pear.
The two initially inked a deal to develop two digital therapeutics for multiple sclerosis and schizophrenia. When the commercialization agreement fell apart, that deal was not mentioned, so it could still be on the table.
Source : mobihealthnews